About Sodium Citrate
All products are for external use only
Free shipping notes: 40 Lb Bag in Box
40 Lb Bag in box ships free in lower 48 continental states. Soapgoods Inc reserves the right to ship free shipping items via any carrier.
Documentation
Identification
- Synonyms: Sodium Citrate, Sodium Citrate Granular
- INCI Name: 6132-04-3
- CAS: 6132-04-3
- Einecs: 612-118-5
The Science
- Viscosity: Solid granular
- Solublity: Soluble in water
- Storage: Cool, dark dry area, air tight container preferred
Characteristics
- Appearance: Fine White Granules
- Odor: Low to no odor
- Natural: Synthetic
- Packaging: 1 Lbs is a single bag, 6 Lb is a single bag, 24 lb is 4 x 6 lb bags, 50 lb is a single 50 lb bag
- Shelf life: Indefinite, Suggested retest date 5 year from purchase
Usage / Benefits
- Industries: Personal care, Cosmetics, Cleaning products, Electroplating, Photography, Textiles, Dairy, Water treatment, Laboratory, Paper industry
- Applications: Buffering agent, Chelating agent, Emulsifying salt, Stabilizer, pH adjustment, Water softener, Coagulation aid, Flavor enhancer, Anticoagulant in blood plasma, Preservative, Textile dyeing, Electrolyte replenishment, Metal cleaning, Complexing agent
- Benefits: Improves texture, Enhances flavor, Stabilizes acidic foods, Prevents crystallization, Enhances solubility, Preserves cosmetics, Prolongs product shelf-life, Improves skin feel in cosmetics, Acts as a natural preservative, Reduces metal ion impact, Facilitates controlled release in products, Aids in pH balance, Enhances product stability.
- Products Uses: Buffering agent, Emulsifier, Anticoagulant in blood transfusions, Acidulant in beverages, Stabilizer in ice cream, Preservative in cosmetics, Chelating agent, Alkalizing agent, Sour salt in culinary applications, Flavor enhancer in confectionery, Softening agent in water treatment.
- Safety: Generally safe, however keep out of eyes and do not eat.
- Cautions: Not for ingestion, keep away from pets and children who may attempt to eat.
- External Use Only: Even if food grade, we do not provide items for ingestion, all of our items are for external use only.
Sodium Citrate
Due to Sodium Citrate being a conjugate base of a weak acid it can be used as a buffering agent which resists changes in pH. Sodium Citrate is used to control acidity in certain substances such as such as gelatin desserts. When antacids such as Alka-Seltzer are dissolved in water Sodium Citrate is the compound that is the product of these substances.
Sodium Citrate is used as an anticoagulant in the preservation and transfusion of blood in blood banks and in blood collection tubes. It is an anticoagulant because the citrate ion chelates the calcium ions in the blood which disrupts the blood clotting mechanism.
Sift the dry ingredients into a bowl and with a wire whisk mix the mixture. Heat some water and place the bottle of oil in the warm water to melt the oil. Carefully and slowly add the oil to the dry ingredients and then add the fragrance. Mix thoroughly. Take 1 tbsp of the mixture and form it with your hands into balls. You can also press them into molds such as the molds used for small chocolate candies. If you do use the molds, put them in the freezer until the bath bombs are hard and can be popped out.
Allow the balls to sit for 2-3 hours on a sheet of waxed paper. Carefully and gently reshape if necessary. Allow the bath bombs to harden and dry out for 24 to 48 hours. Due to their fragility, package each bomb in it's individual closed container or cellophane bag in order to keep them fresh. When ready to take a bath drop one of the bath bombs into warm bath water and it will release oils and fragrance.
Sodium Citrate, a sodium salt of citric acid, is known for its versatile applications, particularly in the personal care, soap making, and cosmetic industries. Its neutralizing and chelating properties make it a sought-after ingredient in various formulations. With its wide-ranging benefits, Sodium Citrate has become a staple in many products that are part of daily life.
Personal Care Products
Within the realm of personal care, Sodium Citrate plays a vital role in enhancing product functionality and user experience. Its applications extend to a multitude of areas:
Stabilization and pH Adjustment
Sodium Citrate acts as a buffering agent, helping to stabilize and adjust the pH level of personal care products. This ensures that products remain gentle on the skin and maintain their efficacy over time.
Chelating Agent
As a chelating agent, Sodium Citrate binds to metal ions, preventing discoloration and rancidity, thus extending the shelf life of products.
Enhancing Sensory Experience
Sodium Citrate contributes to the overall sensory experience, improving the texture and feel of lotions, creams, and other skincare products.
Soap Making
Sodium Citrate is a crucial component in soap making. It offers several benefits that enhance the quality and appeal of handcrafted soaps:
pH Balancing
It helps in maintaining the ideal pH level of soaps, ensuring that they are gentle on the skin.
Water Softening
Sodium Citrate softens water, improving lathering and allowing soaps to rinse off easily without leaving any residue.
Enhanced Transparency
In transparent soap making, Sodium Citrate contributes to clarity, creating aesthetically pleasing transparent soaps.
Cosmetic Making
The cosmetic industry also utilizes Sodium Citrate in various innovative ways:
Texture Enhancer
In cosmetic formulations, Sodium Citrate is used to enhance the texture and spreadability of products such as foundations and creams.
Stabilizing Emulsions
It acts as an emulsion stabilizer, ensuring that oil and water components in creams and lotions remain uniformly mixed.
Preservation
Sodium Citrate's chelating properties also contribute to the preservation of cosmetic products, preventing spoilage and maintaining quality.
Sodium Citrate Uses
Personal Care Products
Sodium Citrate is used to stabilize and adjust the pH level of personal care products, ensuring their gentleness on the skin and extended shelf life.
Soap Making
pH Balancing:
Helps maintain the ideal pH level in soaps.Water Softening:
Acts as a water softener, enhancing lathering and rinsing.Enhancing Transparency:
Contributes to clarity in transparent soaps.
Cosmetic Making
Texture Enhancer:
Used to improve texture and spreadability in cosmetic products.Emulsion Stabilization:
Stabilizes emulsions in creams and lotions.Preservation:
Acts as a chelating agent to preserve quality and prevent spoilage.
Food Industry
Utilized as a flavor enhancer, emulsifier, and preservative in various food products.
Pharmaceutical Industry
Serves as a buffering agent, controlling the pH in medicinal products, and is also used as an anticoagulant in blood collection tubes.
Water Treatment
Employed in water treatment processes to soften water and remove magnesium and calcium ions.
Industrial Applications
Used in various industrial processes, including as a concrete retarder in construction and as a complexing agent in electroplating.
Sodium Citrate Benefits
Buffering Capabilities
Sodium citrate acts as an effective pH buffer, ensuring products maintain a consistent pH level. This stability is essential in many formulations to keep products safe and effective over time.
Emulsifying Properties
Its unique composition enables it to act as an emulsifier, helping in the mixing of oils and water, commonly required in cosmetic and personal care formulations.
Safe Anticoagulant
In medical settings, sodium citrate is utilized as an anticoagulant for blood transfusions and donations, ensuring blood does not clot prematurely.
Flavor Enhancement
Its mild salty and sour taste can enhance the natural flavors of foods and drinks without overpowering them.
Texture Improvement
In the food industry, especially in ice creams and sorbets, sodium citrate can help in achieving a smooth texture by preventing the formation of large ice crystals.
Chelating Abilities
Sodium citrate can bind with metal ions, acting as a chelating agent. This property is particularly beneficial in cosmetics and personal care products where it helps in stabilizing and improving the efficacy of the product.
Preservative Qualities
Its ability to prevent microbial growth makes it a valued preservative in many cosmetic products, prolonging their shelf life.
FAQ
Sodium Citrate Formula
Sodium Citrate is a chemical compound with a formula that reflects its composition:
1. Chemical Composition
The chemical formula for Sodium Citrate is C6H5Na3O7.
2. Molecular Structure
Sodium Citrate consists of carbon atoms (C), hydrogen atoms (H), sodium atoms (Na), and oxygen atoms (O). Its molecular structure includes a chain of citric acid units.
3. Functional Groups
Sodium Citrate contains carboxyl groups and hydroxyl groups, contributing to its properties as a chelating agent and pH regulator.
Applications of Sodium Citrate
Sodium Citrate's formula represents its molecular composition, highlighting its utility as a multifunctional ingredient in various industries beyond the realm of food.
Who is Soapgoods?
We are proud to present a diverse and extensive selection of soap making supplies including soap molds as well as melt and pour soap bases. Are you looking for something unique, something hard to find? Wondering Where to buy Witch Hazel Distillate - Alcohol Free? We are a fantastic source!
We carry it all and many other fantastic but hard to find items at great wholesale prices, we are the one-stop-shop, you can find everything you need here, even bulk Xanthan Gum Right here at Soapgoods
We know you have choices, that's why we work harder, providing better quality, faster shipping and a wide selection of items in practical sizes for any application. When you need to make a purchase and need it delivered quickly, look to us to provide you the service you need. We even have Wholesale Yogurt Powder, in small and large builds sizes, either way, you can get our best direct to consumer pricing. Plus we ship it out the next business days. and will ship the next business day.
Where else can you do all your shopping in one place? When you need to make a purchase and get it delivered quickly. Buy Zinc Oxide Powder? Right here
When you are looking for quality personal care products, cosmetics and soap making supplies, be sure to visit our online store for Acacia Gum Enjoy our premium quality ingredients and super fast shipping times.
Read our Real Reviews from Google and Bizrate
100% Real Reviews (we cannot edit, create or delete reviews) From Google, Click here -> Real SoapGoods Reviews
See more Real Reviews (we cannot edit, create or delete reviews) from Third Party Watchdog Bizrate Real SoapGoods Reviews (Click Here and Scroll Down)
Read hundreds of real testimonials from over a decade of service. SoapGoods Testimonials.
How Fast Can I get it?
We Guarantee Your order ships out the same or next business day! This means in the South East you will have your order in 1 to 3 business days, in the North East normally 3 to 4 days and in the West normally 4 to 5 days. For full details on shipping and processing times please see our expected delivery times.
These times are based on business days, not including weekends or holidays.
FedEx Delivery Map
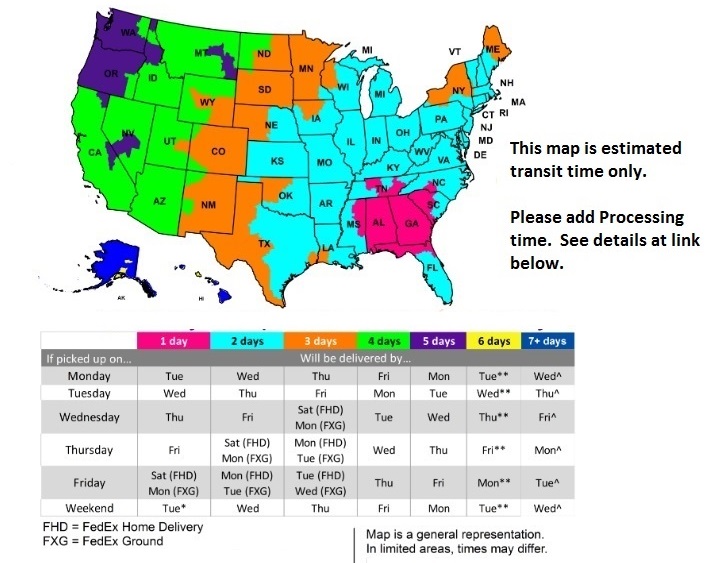
For Processing Times Click Here
USPS Delivery Map
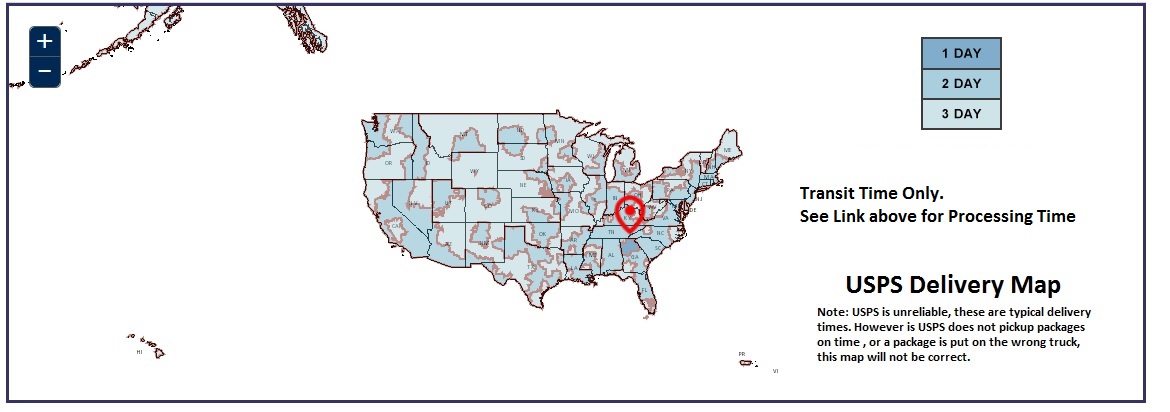
Typical Delivery Times to Major US Cities
| Major Cities | Total Business Days +1 / -1 |
|---|---|
| Alabama (AL) - Montgomery, Birmingham | 1 |
| Alaska (AK) - Juneau, Anchorage | 7 |
| Arizona (AZ) - Phoenix, Tucson | 4 |
| Arkansas (AR) - Little Rock, Fayetteville | 2 |
| California (CA) - Sacramento, Los Angeles, San Francisco, San Diego, Sacramento, San Jose | 4 |
| Colorado (CO) - Denver, Colorado Springs | 3 |
| Connecticut (CT) - Hartford, New Haven | 2 |
| Delaware (DE) - Dover, Wilmington, Newark | 2 |
| Florida (FL) - Tallahassee, Orlando, Miami, Jacksonville, Tampa, Destin | 2 |
| Georgia (GA) - Atlanta, Savannah, Augusta, Athens | 1 |
| Hawaii (HI) - Honolulu, Kailua | 7 |
| Idaho (ID) - Boise, Coeur d'Alene | 4 |
| Illinois (IL) - Springfield, Chicago, Peoria, Rockford | 2 |
| Indiana (IN) - Indianapolis, Fort Wayne | 2 |
| Iowa (IA) - Des Moines, Cedar Rapids | 2 |
| Kansas (KS) - Topeka, Wichita, Kansas City | 2 |
| Kentucky (KY) - Frankfort, Louisville, Lexington | 2 |
| Louisiana (LA) - Baton Rouge, New Orleans, Lafayette | 2 |
| Maine (ME) - Augusta, Portland, Bangor | 3 |
| Maryland (MD) - Annapolis, Baltimore | 2 |
| Massachusetts (MA) - Boston, Cambridge, Worcester | 2 |
| Michigan (MI) - Lansing, Detroit, Grand Rapids | 2 |
| Minnesota (MN) - St. Paul, Minneapolis, Duluth | 3 |
| Mississippi (MS) - Jackson, Biloxi, Hattiesburg | 1 |
| Missouri (MO) - Jefferson City, St Louis, Kansas City | 2 |
| Montana (MT) - Helena, Billings | 4 |
| Nebraska (NE) - Lincoln, Omaha | 2 |
| Nevada (NV) - Carson City, Las Vegas, Reno | 4 |
| New Hampshire (NH) - Concord, Manchester, Portsmouth | 2 |
| New Jersey (NJ) - Trenton, Newark, Jersey City | 2 |
| New Mexico (NM) - Santa Fe, Alburquerque | 3 |
| New York (NY) - Albany, New York, Rochester, Buffalo, Albany, Syracuse, Niagara Falls, Ithaca | 3 |
| North Carolina (NC) - Raleigh, Charlotte | 2 |
| North Dakota (ND) - Bismarck, Fargo | 3 |
| Ohio (OH) - Columbus, Cleveland, Cincinnati | 2 |
| Oklahoma (OK) - Oklahoma City, Fairview, | 2 |
| Oregon (OR) - Salem, Portland, Eugene | 5 |
| Pennsylvania (PA) - Harrisburg, Philadelphia, Pittsburgh | 2 |
| Rhode Island (RI) - Providence, Newport | 2 |
| South Carolina (SC) - Columbia, Charleston | 1 |
| South Dakota (SD) - Pierre, Sioux Falls, Rapid City | 3 |
| Tennessee (TN) - Nashville, Memphis | 2 |
| Texas (TX) - Austin, Houston, Dallas | 3 |
| Utah (UT) - Salt Lake City, St. George | 3 |
| Vermont (VT) - Montpelier, Burlington | 3 |
| Virginia (VA) - Richmond, Virginia Beach | 2 |
| Washington (WA) - Olympia, Seattle, Vancouver, Spokane | 5 |
| West Virginia (WV) - Charleston, Morgantown | 2 |
| Wisconsin (WI) - Madison, Milwaukee | 2 |
| Wyoming (WY) - Cheyenne, Jackson | 4 |
Disclaimer: All product descriptions and specifications provided in this description are intended as a guide only and are subject to change without notice. While we strive for accuracy, discrepancies or errors may be present. These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease.
No posts found











